HIPAA now has stricter and more explicit requirements. Especially as enforcement expectations tighten. This is changing how medical practices and business associates operate day to day. The big shift is that “good faith” compliance is no longer enough. Regulators now expect documented and continuously maintained compliance.
Compliance Must Be Documented, Not Assumed
Organizations can no longer rely on informal policies, verbal training, or “we’ve always done it this way.”
Written risk analyses, risk management plans, and policies have always been required. But now, regulators are closely reviewing for updates. Documents must be current, not created once and forgotten.
If it’s not documented, Office for Civil Rights treats it as if it doesn’t exist.
Impact: More time spent maintaining documentation, but far less exposure during an audit or complaint.
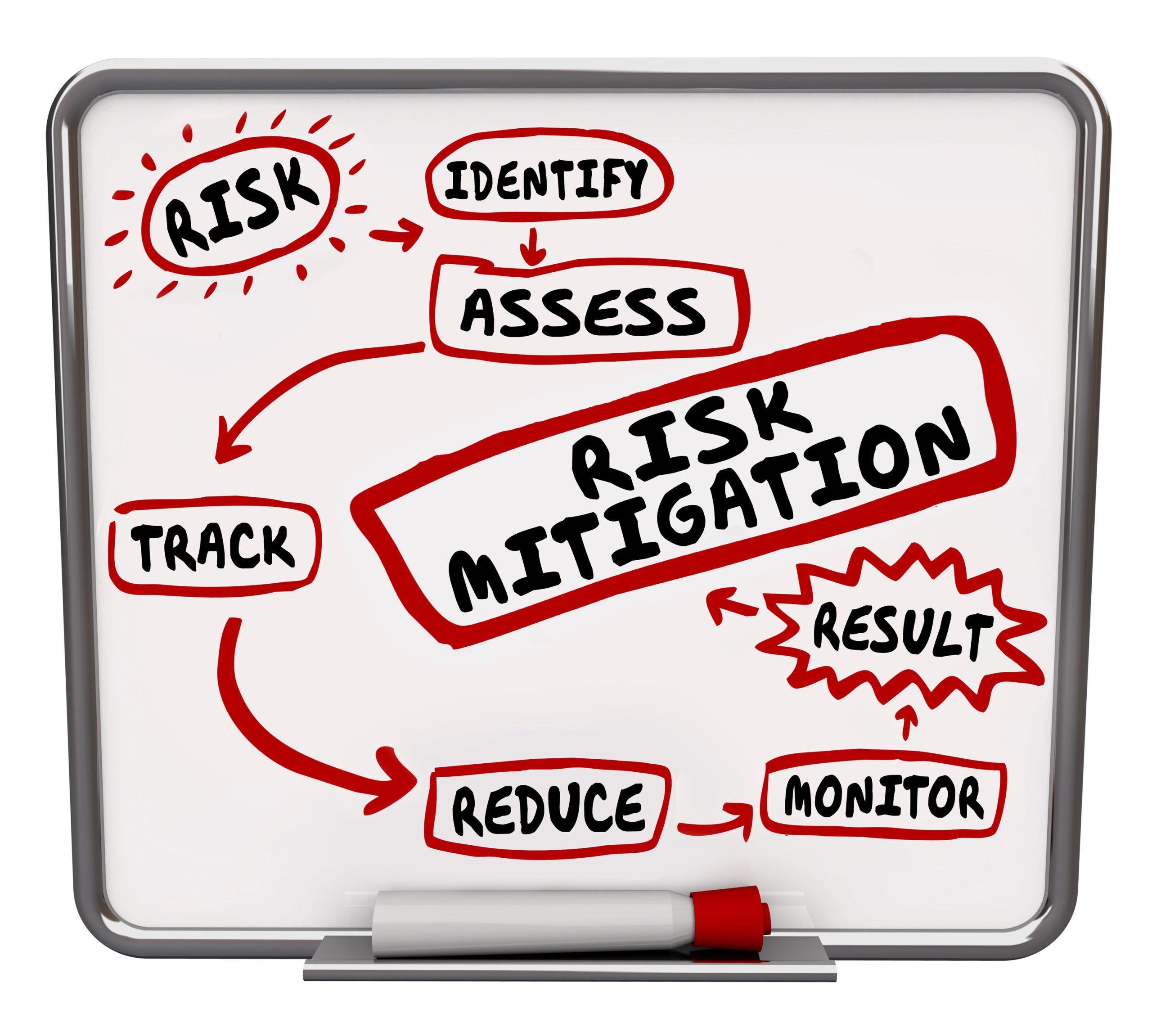
Risk Analysis Is the Foundation of Everything
The Office for Civil Rights (OCR) has made it crystal clear that risk analysis drives compliance decisions. Security controls must align with identified risks. Then a documented risk management plan that outlines the mitigation process must be created. “Addressable” safeguards must be justified if not implemented, this was never meant to be optional! Generic or copied risk analyses are being rejected.
Impact: Organizations must understand their systems, vendors, workflows, and vulnerabilities – not someone else’s.
Cybersecurity Expectations Are Higher
HIPAA now expects organizations to adopt modern security practices, not outdated basics.
- Multi-factor authentication (MFA)
- Encryption of data at rest and in transit
- Regular patching and system hardening
- Monitoring for suspicious activity
Failing to implement common-sense safeguards is increasingly viewed as willful neglect.
Impact: Greater reliance on IT partners, but also more oversight and accountability.
Vendors and Business Associates Are Under a Microscope
Practices are responsible for who they share PHI with. Business Associate Agreements (BAAs) must be current. Business associates must have current subcontractor agreements in place as well. Vendors must demonstrate their own security practices and comply with the HIPAA rules. “We trusted our vendor” is no longer a defense. Covered entities are responsible for ensuring their vendors are compliant.
Impact: More vendor vetting, more paperwork, fewer risky shortcuts.
Training Must Be Ongoing
Annual, generic HIPAA training doesn’t cut it anymore. Training must address phishing, ransomware, and real-world threats. Training must be tracked and documented.
Impact: Better-informed staff equals fewer costly human-error breaches.
Faster Response and Accountability After Incidents
HIPAA enforcement now scrutinizes how quickly and effectively a practice responds to incidents. Incident response plans must exist before an event occurs. Delays or confusion during a breach increases penalties. Internal security incident investigations must be documented.
Impact: Organizations need clear procedures, not panic, when something goes wrong.
Small Practices Are No Longer “Too Small to Enforce”
Enforcement actions increasingly involve:
- Small and solo practices
- Dental offices
- Specialty clinics
- Business associates
Complaints, not breaches often trigger investigations.
Impact: Every organization is expected to meet the same baseline standards, regardless of size.
Summary
HIPAA’s stricter requirements mean organizations must shift from reactive compliance to ongoing risk management.
Aris Medical Solutions helps medical practices and business associates understand HIPAA expectations and reduce risk- step by step.
Our HIPAA Keeper™ was designed to help organizations:
- Understand where they stand
- Organize required documentation
- Maintain compliance over time
- Be prepared if questions ever arise
Additionally, you will have a HIPAA security analyst to guide and assist you when you need help.

To find out where you stand with your compliance, schedule a free HIPAA checkup today at Aris Medical Solutions.